Genetic Extinction Technology
(Biological weapon/Research, research, Genetic engineering) | |
|---|---|
| Interest of | Bill Gates |
| genetic engineering technology that the official narrative claims is able to propagate a particular suite of genes throughout a population | |
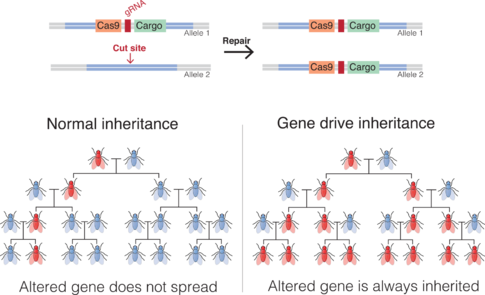
A gene drive is an technology of genetic engineering that the official narrative claims is able to propagate a particular suite of genes throughout a population[1] by altering the probability that a specific allele will be transmitted to offspring (instead of the Mendelian 50% probability). It application is particularly suited for creating an irreversible species extinction[2].
The U.S. Defense Advanced Research Projects Agency (DARPA) has given approximately $100 million for gene drive research, making them likely the largest single funder of gene drive research on the planet. The secretive top-level JASON group of military advisors produced a classified study on gene drive in 2017, reflecting an extremely high level of interest and activity by other sections of the U.S. military and Intelligence community[3].
The Bill and Melinda Gates Foundation paid a PR firm $1.6 million to secretly stack key UN advisory processes with gene drive-friendly scientists[4].
A Powerful Technique
Gene drives can arise through a variety of mechanisms.[5][6] They have been proposed to provide an effective means of genetically modifying specific populations and entire species.
The technique can employ adding, deleting, disrupting, or modifying genes.[7][8]
Proposed applications include exterminating insects that carry pathogens (notably mosquitoes that transmit malaria, dengue, and zika pathogens), controlling invasive species, or eliminating herbicide or pesticide resistance.[9][10][11][12]
As with any potentially powerful technique, gene drives can be misused in a variety of ways or induce unintended consequences. For example, a gene drive intended to affect only a local population might spread across an entire species. Gene drives used to eradicate populations of invasive species in their non-native habitats may have consequences for the population of the species as a whole, even in its native habitat. Any accidental return of individuals of the species to its original habitats, through natural migration, environmental disruption (storms, floods, etc.), accidental human transportation, or purposeful relocation, could unintentionally drive the species to extinction if the relocated individuals carried harmful gene drives.[13]
“It is very much easier to kill or sterilise a plant using gene editing than it is to make it herbicide or insect-resistant.”
Guy Reeves, expert in GM insects at the Max Planck Institute for Evolutionary Biology (2018) [14]
Gene drives can be built from many naturally occurring selfish genetic elements that use a variety of molecular mechanisms.[15] These naturally occurring mechanisms induce similar segregation distortion in the wild, arising when alleles evolve molecular mechanisms that give them a transmission chance greater than the normal 50%.
Most gene drives have been developed in insects, notably mosquitoes, as a way to control insect-borne pathogens. Recent developments designed gene drives directly in viruses, notably herpesviruses. These viral gene drives can propagate a modification into the population of viruses, and aim to reduce the infectivity of the virus.[16][17]
Interested parties
Defense Advanced Research Projects Agency
In December 2017, documents released under the US Freedom of Information Act showed that the military research agency DARPA had invested $100 million in gene drive research,[18], making them likely the largest single funder of gene drive research on the planet. The documents also reveal that DARPA either funds or co-ordinates with almost all major players working on gene drive development as well as the key holders of patents on CRISPR gene editing technology.
“Given that Darpa is a military agency, we find it surprising that the obvious and concerning dual-use aspects of this research have received so little attention,”
Felix Beck - lawyer at the University of Freiburg (2018) [14]
JASON Military Advisory Group
The secretive top-level JASON group of military advisors produced a classified study on gene drive in 2017, reflecting an extremely high level of interest and activity by other sections of the U.S. military and Intelligence community[19].
Emails show that the JASON study was initiated with a two day meeting of a select group of invited gene drive researchers in June 2017. At the meeting, the VP of Global Biotechnology for Monsanto gave a presentation on crop science and gene drives[20].
The Intelligence Advanced Research Projects Agency
The Intelligence Advanced Research Projects Agency (IARPA), an organization within the Office of the Director of National Intelligence, has also expressed interest in funding gene drive work. A scientist involved describes IARPA as “basically the intelligence agencies version of DARPA, which may be more frightening"[21]
Project for the New American Century
In their 2000 paper: "Rebuilding America's Defences: Strategies, Forces And Resources For A New Century", PNAC commented on biowarfare application of a then unnamed technology:
“And advanced forms of biological warfare that can “target” specific genotypes may transform biological warfare from the realm of terror to a politically useful tool.”
Project for the New American Century [22]
Bill & Melinda Gates Foundation
The Bill and Melinda Gates Foundation paid a PR firm $1.6 million to secretly stack key UN advisory processes with gene drive-friendly scientists, and recruited ostensibly independent academics and public officials into a private collaboration to counteract proposed regulations and to resist calls by scientists and conservationists for an international moratorium[23].
Target Malaria, a project funded by the Bill and Melinda Gates Foundation, invested $75 million in gene drive technology. The foundation originally estimated the technology to be ready for field use by 2029 somewhere in Africa. However, in 2016 Gates changed this estimate to some time within the following two years.[24]
Because Target Malaria hopes to deploy their gene drives in African countries they have been at pains to emphasize independence from military agendas. However,Target Malaria’s Andrea Crisanti (working at Imperial College) is also a lead grantee or subcontractor for DARPA’s Safe Genes project, having confirmed he has been hired by DARPA on a $2.5m contract[25].
Imperial College London
Imperial College London has been a pioneer in gene drive research, with DARPA funding[26]. In trials 2016-2018, scientists succeeded in destroying a population of mosquitoes in a lab by introducing a genetic mutation that spread through the population and eventually sterilized all of the mosquitoes. In previous experiments, mosquitoes had small random mutations that immunized them against the gene drive. The Imperial College scientists created a gene drive that did not fall prey to this type of resistance.[27]
Related Quotations
| Page | Quote | Author | Date |
|---|---|---|---|
| Biological weapon | “Fabricating scary narratives about superbugs is much easier than delivering on promises of making those bugs in labs. This is also a very productive avenue as people are woefully gullible and thus can be controlled by narratives just as effectively as by an actual scary-scary bioengineered virus. Biodefense is a huge grift on both sides. 'Their' side appropriates money and power, and new billion dollar agencies for 'Pandemic Preparedness'. 'Our' side gets millions of followers talking about them evil guys, or spinning stories about biolabs in Wuhan, Ukraine and lately California. They leak' from labs almost every week, and using CRISPER gene drive narrative logic, all mice in the world should look like Ralph Baric by now.” | Sasha Latypova | November 2023 |
| Transhumanism | “Even if half the world’s species were lost [during genetic experiments], enormous diversity would still remain. When those in the distant future look back on this period of history, they will likely see it not as the era when the natural environment was impoverished, but as the age when a plethora of new forms—some biological, some technological, some a combination of the two—burst onto the scene. We best serve ourselves, as well as future generations, by focusing on the short-term consequences of our actions rather than our vague notions about the needs of the distant future.” | Gregory Stock | 1993 |
Sponsor
| Event | Description |
|---|---|
| Open Philanthropy | Grant maker funneling deep state money among other things to pandemic planning. Financed Event 201. |
References
- ↑ >https://www.nature.com/news/us-defence-agencies-grapple-with-gene-drives-1.22345
- ↑ https://geneticliteracyproject.org/2020/08/18/viewpoint-is-there-a-scientific-basis-to-ban-gene-drive-technology-that-can-rid-us-of-virus-carrying-rodents-and-mosquitoes/
- ↑ http://genedrivefiles.synbiowatch.org/2017/12/01/us-military-gene-drive-development/
- ↑ https://etcgroup.org/content/gene-drive-files
- ↑ https://doi.org/10.1038%2Fnrg.2015.34
- ↑ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6195636
- ↑ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4117217
- ↑ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1691325
- ↑ http://news.sciencemag.org/biology/2014/07/u-s-researchers-call-greater-oversight-powerful-genetic-technology%7Caccess-date=2014-07-18
- ↑ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4117217
- ↑ https://doi.org/10.1089%2Fvbz.2007.0273
- ↑ https://portals.iucn.org/library/node/48408
- ↑ https://www.wired.com/story/this-gene-editing-tech-might-be-too-dangerous-to-unleash/
- ↑ Jump up to: a b https://www.independent.co.uk/news/science/us-military-plan-biological-weapons-insect-allies-virus-crop-darpa-a8568996.html
- ↑ Champer J, Buchman A, Akbari OS, "Cheating evolution: engineering gene drives to manipulate the fate of wild populations", Nature Reviews. Genetics, volume 17, issue 3, pages 146–59, March 2016, doi = 10.1038/nrg.2015.34
- ↑ https://www.nature.com/articles/s41467-020-18678-0
- ↑ https://www.acsh.org/news/2020/09/30/gene-drives-could-kill-mosquitoes-and-suppress-herpesvirus-infections-15060
- ↑ https://www.theguardian.com/science/2017/dec/04/us-military-agency-invests-100m-in-genetic-extinction-technologies
- ↑ http://genedrivefiles.synbiowatch.org/2017/12/01/us-military-gene-drive-development/
- ↑ http://genedrivefiles.synbiowatch.org/2017/12/01/us-military-gene-drive-development/
- ↑ http://genedrivefiles.synbiowatch.org/20170716-fw_-gbird-update-engagements-and-other-important-items-472/
- ↑ https://web.archive.org/web/20020923154604/http://www.newamericancentury.org/RebuildingAmericasDefenses.pdf
- ↑ https://etcgroup.org/content/gene-drive-files
- ↑ https://www.technologyreview.com/s/602304/bill-gates-doubles-his-bet-on-wiping-out-mosquitoes-with-gene-editing/
- ↑ https://www.theguardian.com/science/2017/dec/04/us-military-agency-invests-100m-in-genetic-extinction-technologies
- ↑ https://www.theguardian.com/science/2017/dec/04/us-military-agency-invests-100m-in-genetic-extinction-technologies
- ↑ https://www.statnews.com/2018/09/24/gene-drive-lab-mosquitos/